Drawing Of The Reaction Of Hydrochloric Acid With Water
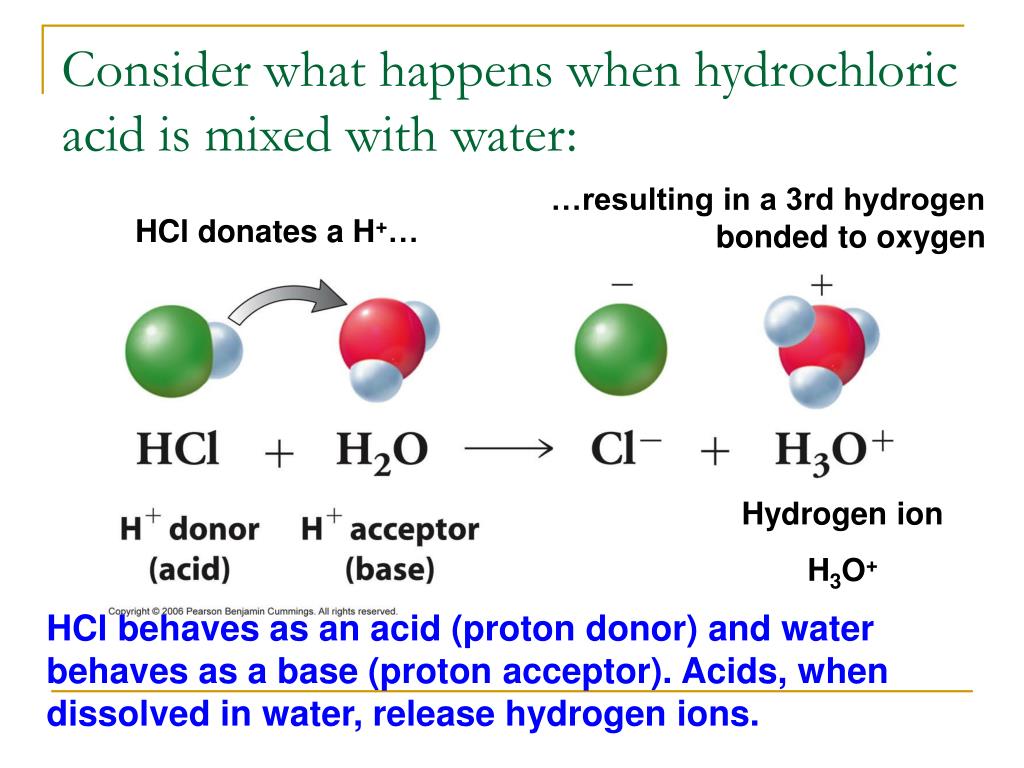
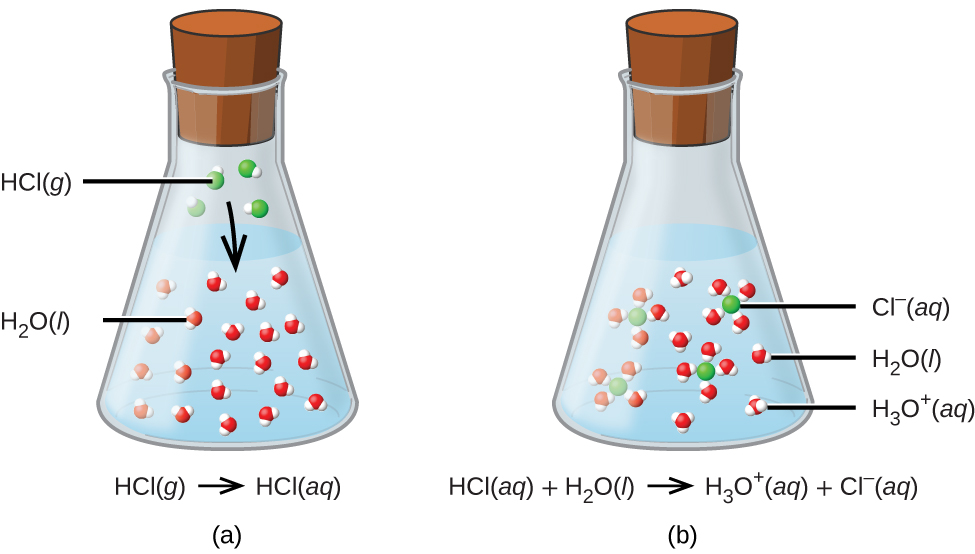
Drawing Of The Reaction Of Hydrochloric Acid With Water - Hydrochloric acid, hcl, is a strong acid, so right from the start you should expect it to ionize completely in aqueous solution. Acid plus base yields water plus salt. Through a process known as hydrolysis, the ions produced when an acid and base combine may react with the water molecules to produce a solution that is slightly acidic or basic. Hydrochloric acid is a strong acid which dissociates into h + and cl − ions in water. Use appropriate tools to draw a schematic representation of the products, showing the hydrated ions and water molecule orientation. Hcl (aq) ⇌ h + (aq) + cl − (aq) This process is a highly exothermic reaction. If the base is a metal hydroxide, then the general formula for the reaction of an acid with a base is described as follows: In this video we will look at the equation for hcl + h2o and write the products. In the laboratory preparation of hydrochloric acid, hydrogen chloride gas is dissolved in water. Hydrochloric acid is a strong acid which dissociates into h + and cl − ions in water. Even in the absence of water, hydrogen chloride can still act as an acid. Use appropriate tools to draw a schematic representation of the products, showing the hydrated ions and water molecule orientation. In this reaction, a proton is transferred from hcl (the acid, or proton donor ) to hydroxide ion (the base, or proton acceptor ). Hydrochloric acid is prepared by dissolving gaseous hydrogen chloride in water. Hydrochloric acid is a strong acid, stronger than water, so it’ll force water to act as a base instead. Give two reasons for the same. Hydrochloric acid, hcl, is a strong acid, so right from the start you should expect it to ionize completely in aqueous solution. Here’s the best way to solve it. [ (% × d) / mw] × 10 = molarity. Includes kit list and safety instructions. The following equation is used for calculating acid and base molarity where the concentration is given in wt %: (ii) why is such an arrangement necessary? Through a process known as hydrolysis, the ions produced when an acid and base combine may react with the water molecules to produce a solution that is slightly. Use appropriate tools to draw a schematic representation of the products, showing the hydrated ions and water molecule orientation. D = density (or specific gravity); For example, the reaction of equimolar amounts of hbr and naoh to give water and a salt (nabr) is a neutralization reaction: Hydrochloric acid is a strong acid, stronger than water, so it’ll force water. The ionization of hydrochloric acid in water is given below: For example, hydrochloric acid, hcl, as a strong acid it donates a proton to water, h2o, to form the hydronium ion, h3o plus, and the conjugate base to hcl which is the chloride anion, cl minus. This process is a highly exothermic reaction. Since the h+ (often called a “proton”). Through a process known as hydrolysis, the ions produced when an acid and base combine may react with the water molecules to produce a solution that is slightly acidic or basic. D = density (or specific gravity); Since the h+ (often called a “proton”) and. As a general concept, if a strong acid is mixed with a weak base, the. A class practical on reacting magnesium with hydrochloric acid and how to measure the rate of reaction. The reaction takes place as follows: If you paired water with something which is a weaker acid than water, then water will act as an acid. The above equation can then be used to calculate the molarity of the 70 wt % nitric. The water dissociation constant remains the same whether the aqueous solution is neutral, acidic, or basic, i.e.: In reality, this reaction reaches an equilibrium. [ (% × d) / mw] × 10 = molarity. The following equation is used for calculating acid and base molarity where the concentration is given in wt %: Acid plus base yields water plus salt. The following equation is used for calculating acid and base molarity where the concentration is given in wt %: The water dissociation constant remains the same whether the aqueous solution is neutral, acidic, or basic, i.e.: In this video we will look at the equation for hcl + h2o and write the products. The reaction takes place as follows: Hcl(aq). Hydrochloric acid is a strong acid which dissociates into h + and cl − ions in water. The reaction takes place as follows: Hydrochloric acid + magnesium →. The water dissociation constant remains the same whether the aqueous solution is neutral, acidic, or basic, i.e.: (ii) why is such an arrangement necessary? Acid + metal → salt + hydrogen. Hydrochloric acid, hcl, is a strong acid, so right from the start you should expect it to ionize completely in aqueous solution. Hcl(aq) hx+(aq) +clx−(aq) h c l ( a q) h x + ( a q) + c l x − ( a q) i understand that when added to water the. In reality, this reaction reaches an equilibrium. The acid dissociation or ionization constant, k a, is large, which means hcl dissociates or ionizes practically completely in water. The water dissociation constant remains the same whether the aqueous solution is neutral, acidic, or basic, i.e.: The above equation can then be used to calculate the molarity of the 70 wt %. The resulting solution is called hydrochloric acid and is a strong acid. For example, the reaction of equimolar amounts of hbr and naoh to give water and a salt (nabr) is a neutralization reaction: Give two reasons for the same. Includes kit list and safety instructions. (i) draw a diagram to show the arrangement used for the absorption of hcl in water. The acid dissociation or ionization constant, k a, is large, which means hcl dissociates or ionizes practically completely in water. Acid + metal → salt + hydrogen. For example, hydrochloric acid, hcl, as a strong acid it donates a proton to water, h2o, to form the hydronium ion, h3o plus, and the conjugate base to hcl which is the chloride anion, cl minus. [ (% × d) / mw] × 10 = molarity. In the laboratory preparation of hydrochloric acid, hydrogen chloride gas is dissolved in water. (iii) write the chemical equation for the laboratory preparation of hcl gas when the reactants are: (ii) why is such an arrangement necessary? Even in the absence of water, hydrogen chloride can still act as an acid. Hydrochloric acid is prepared by dissolving gaseous hydrogen chloride in water. Use appropriate tools to draw a schematic representation of the products, showing the hydrated ions and water molecule orientation. The ionization of hydrochloric acid in water is given below:PPT Unit 7 Acids and Bases PowerPoint Presentation, free download
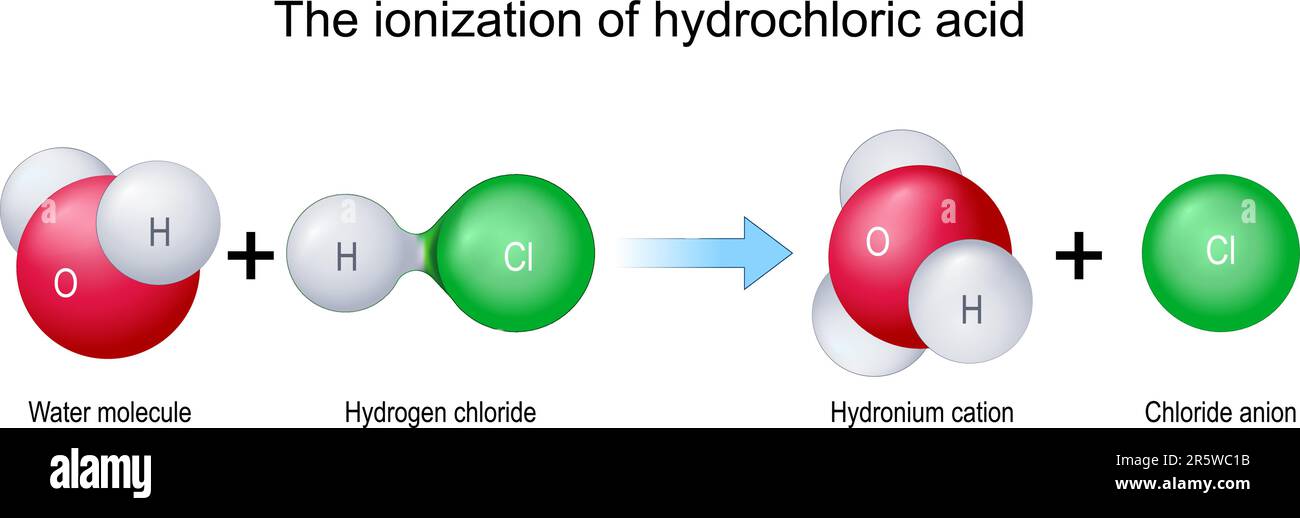
Chemical Equation For The Acid Ionization Of Hydrochloric Hcl In Water
Complete The Chemical Equation For Acid Ionization Of Hydrochloric Hcl
Reaction of Hydrochloric Acid with Water, Chemistry Lecture Sabaq.pk
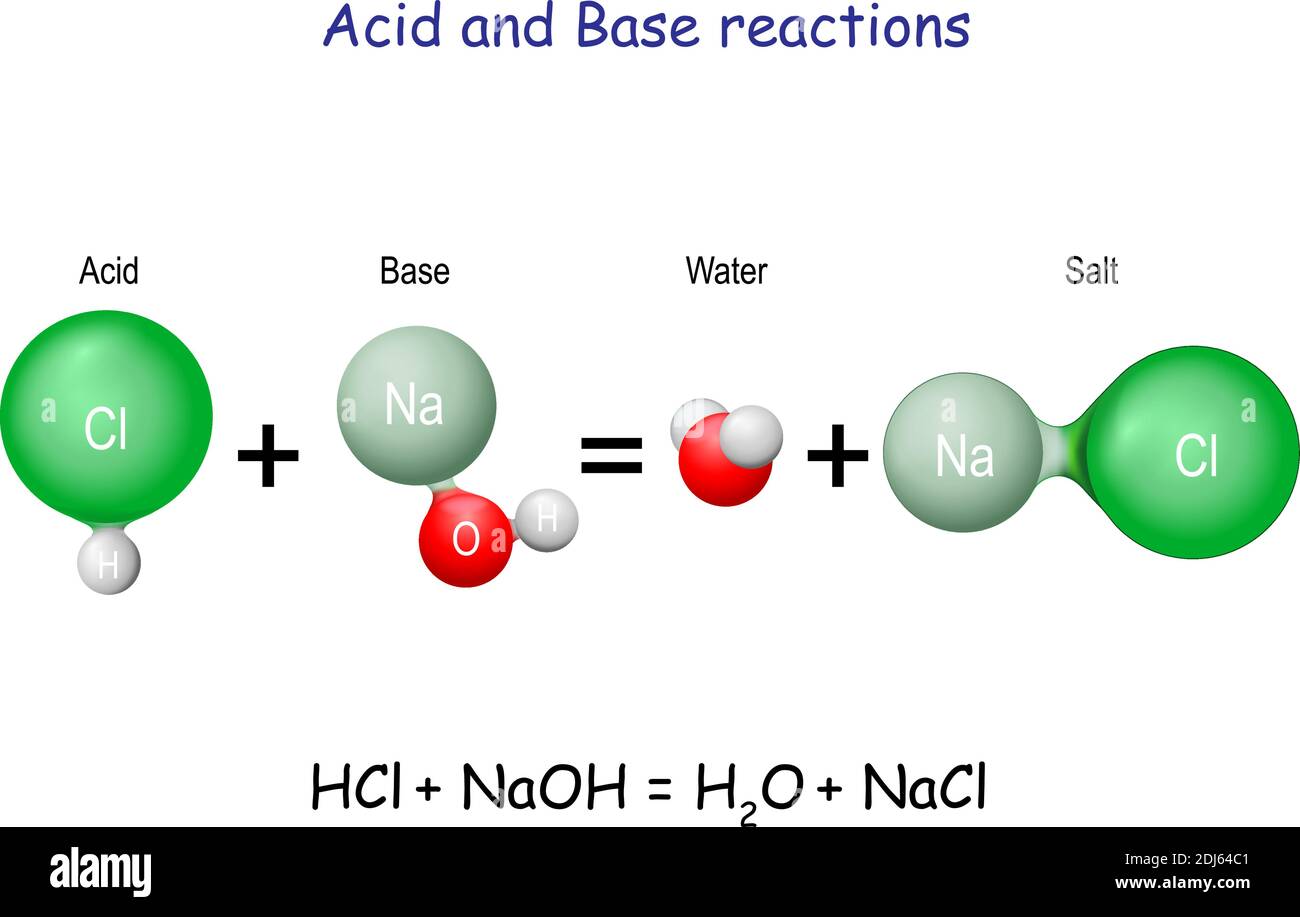
4.3 AcidBase Reactions Introduction to Chemistry
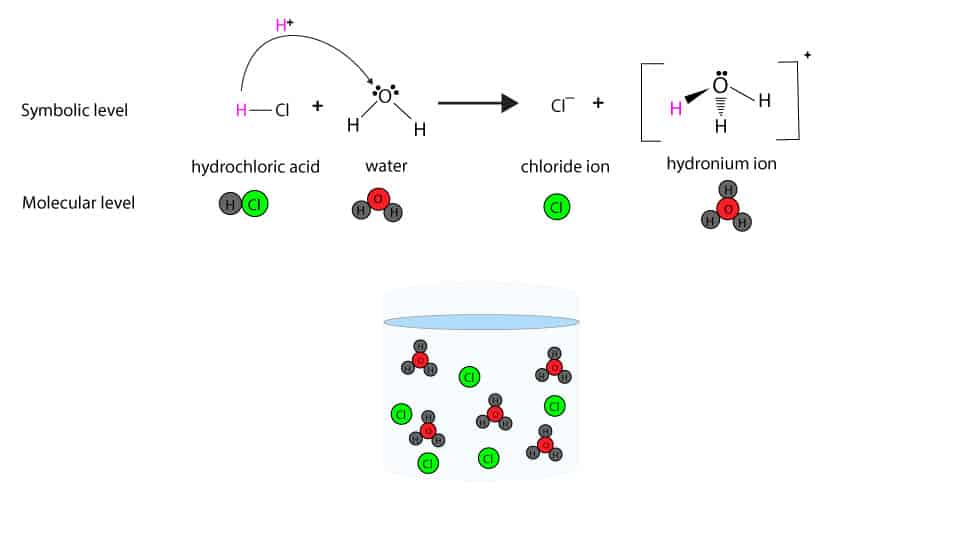
The ionization of hydrochloric acid. Molecules H2O and HCl combine to
What is the chemical equation for HCl dissolving into water and
Hydrochloric acid molecule Stock Vector Images Alamy
[DIAGRAM] Phase Diagram Hcl Water
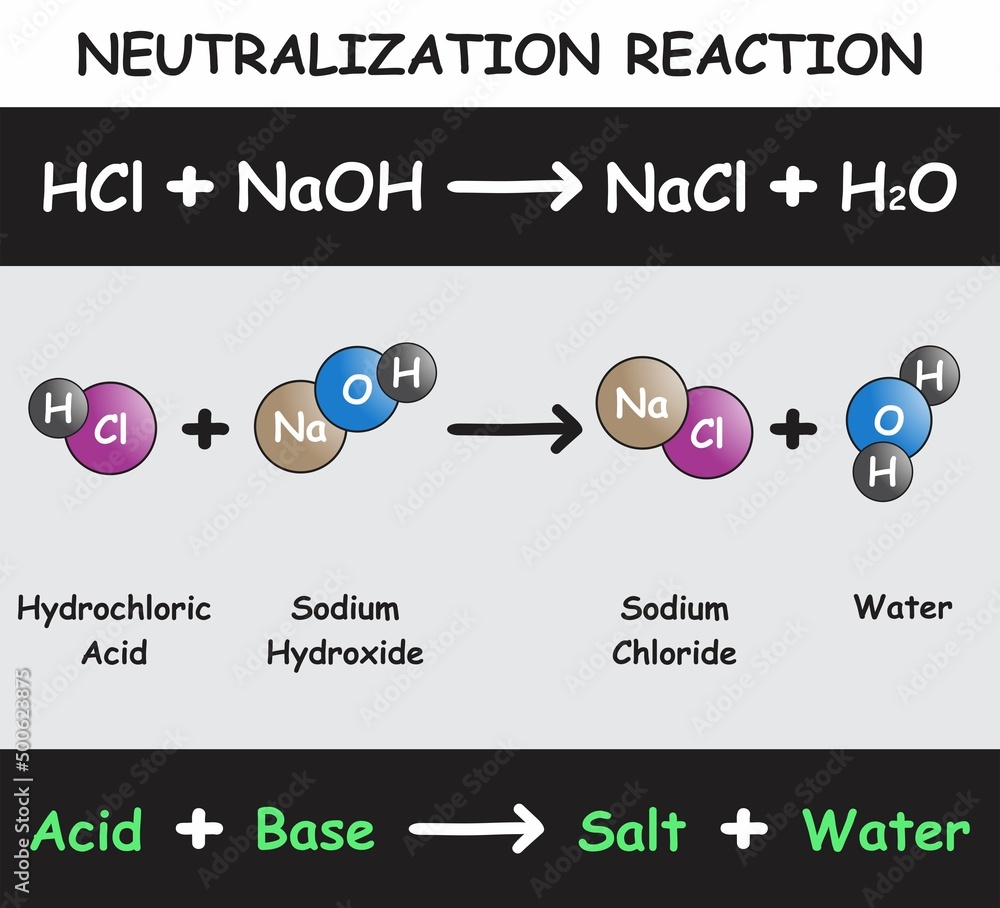
Neutralization Reaction Infographic Diagram with example of
Hydrochloric Acid, Hcl, Is A Strong Acid, So Right From The Start You Should Expect It To Ionize Completely In Aqueous Solution.
Hydrochloric Acid Is A Strong Acid Which Dissociates Into H + And Cl − Ions In Water.
Draw The Structures Of Methanol (\(\Mathrm{Ch}_{3}\Mathrm{Oh}\)), Acetic Acid (\(\Mathrm{Ch}_{3}\Mathrm{Cooh}\)), And Methane (\(\Mathrm{Ch}_{4}\)) And Write A Potential Reaction With Water.
As A General Concept, If A Strong Acid Is Mixed With A Weak Base, The Resulting Solution Will Be Slightly Acidic.
Related Post:
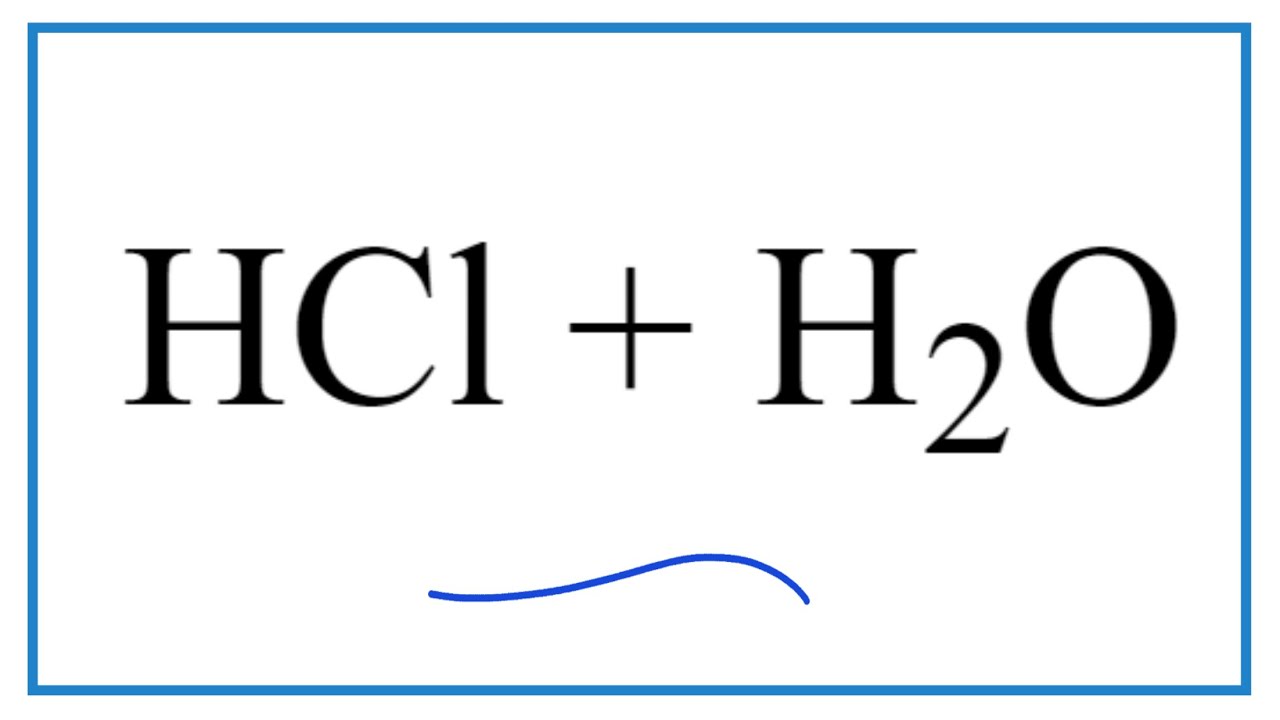

![[DIAGRAM] Phase Diagram Hcl Water](http://c8.alamy.com/comp/H3N06T/diagram-of-the-laboratory-preparation-of-carbon-dioxide-from-hydrochloric-H3N06T.jpg)